The findings might possibly enhance the success rate of cancer drug advancement.
Around 90% of drugs do not reach the marketplace, highlighting the clear requirement for increased effectiveness in drug advancement. The story isn’t various for drugs focused on dealing with cancer, with lots of stopping working due to numerous factors. Now, scientists have actually exposed one reason that particular anti-cancer substances can trigger unforeseen adverse effects. This research study might assist direct an understanding of why some drugs reveal more guarantee than others, supplying a brand-new tool that can be utilized to recognize those drugs and drug prospects.
Among the most necessary and energy-consuming cellular procedures is ribosome biogenesis, the development of the cellular devices that produce all proteins. For cancer cells, this procedure is vital. A current research study released in the journal eLife from the Stowers Institute for Medical Research study evaluated over 1,000 existing anti-cancer drugs to evaluate how they affect the structure and function of the nucleolus, the common cellular organelle where ribosomes are made.
” All cells need to make proteins to work, so they need to make ribosomes, which are likewise protein complexes themselves,” stated lead author Tamara Potapova, Ph.D., a research study professional in the laboratory of Detective Jennifer Gerton, Ph.D. “In cancer cells, ribosome production need to remain in overdrive to make up for high expansion rates needing a lot more proteins.”
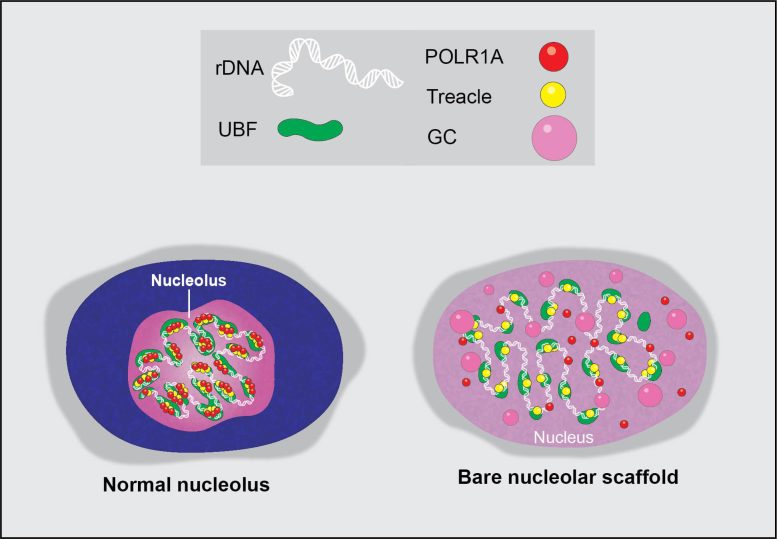
Visual illustration of a regular nucleolus and its severe tension state following transcriptional cyclin-dependent kinase inhibition by chemotherapy representatives. Credit: Image thanks to Mark Miller and Tamara Potapova, Stowers Institute for Medical Research Study
The nucleolus is an unique part of the cell nucleus that houses ribosomal DNA, and where ribosomal RNA production and ribosome assembly mostly happens. Nucleoli can differ significantly in look, functioning as visual signs of the general health of this procedure. Therefore, the group discovered a method to profit from this variation and asked how chemotherapy drugs affect the nucleolus, triggering nucleolar tension.
” In this research study, we not just assessed how anti-cancer drugs modify the look of nucleoli however likewise determined classifications of drugs that trigger unique nucleolar shapes,” stated Gerton. “This allowed us to produce a category system for nucleoli based upon their look that is a resource other scientists can utilize.”
Since cancer’s trademark is unattended expansion, the majority of existing chemotherapeutic representatives are developed to slow this down. “The reasoning was to see whether these drugs, deliberately or accidentally, are impacting ribosome biogenesis and to what degree,” stated Potapova. “Striking ribosome biogenesis might be a double-edged sword– it would hinder the practicality of cancer cells while all at once modifying protein production in typical cells.”
Various drugs affect various paths associated with cancer development. Those that affect ribosome production can cause unique states of nucleolar tension that manifest in quickly seen morphological modifications. Nevertheless, nucleolar tension can be tough to determine.
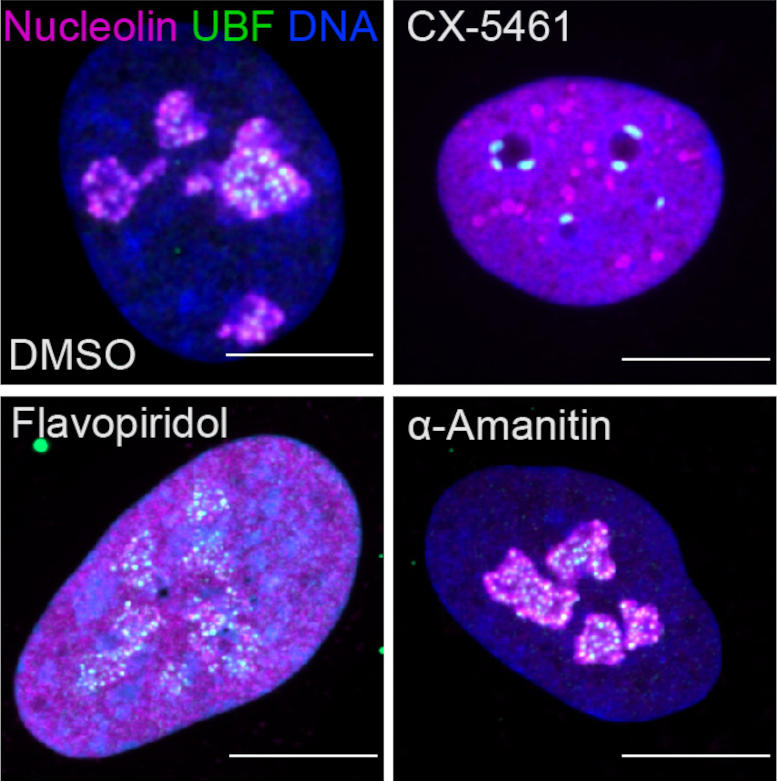
Fluorescent images revealing nucleolar tension caused by drugs that prevent transcriptional enzymes, or cyclin-dependent kinases (CDK). The upper left panel reveals a regular cell with 2 essential nucleolar proteins stained (magenta and green) and DNA (blue). The staying panels reveal the effect of CDK or transcription-inhibitory drugs on nucleoli. Credit: Image thanks to Tamara Potapova, Gerton Laboratory, Stowers Institute for Medical Research Study
” This was among the concerns that hindered this field,” stated Potapova. “Cells can have various varieties of nucleoli with various shapes and sizes, and it has actually been challenging to discover a single criterion that can totally explain a “typical” nucleolus. Establishing this tool, which we described “nucleolar normality rating,” enabled us to determine nucleolar tension specifically, and it can be utilized by other laboratories to determine nucleolar tension in their speculative designs.”
Through the thorough screening of anti-cancer substances on nucleolar tension, the group determined one class of enzymes in specific, cyclin-dependent kinases, whose inhibition ruins the nucleolus practically entirely. A number of these inhibitors stopped working in scientific trials, and their damaging influence on the nucleolus was not totally valued formerly.
Drugs typically stop working in scientific trials due to extreme and unexpected toxicity that can be brought on by their off-target impacts. This indicates that a particle developed to target one path might likewise be affecting a various path or preventing an enzyme needed for cellular function. In this research study, the group discovered a result on a whole organelle.
” I hope at a minimum this research study increases awareness that some anti-cancer drugs can trigger unexpected interruption of the nucleolus, which can be extremely popular,” stated Potapova. “This possibility needs to be thought about throughout brand-new drug advancement.”